I’m very interested in contrails, but when you look for information about them on the internet, half the sites that turn up are about a conspiracy theory which claims that any trail that last more than a few minutes is actually a “chemtrail”, comprised of dangerous chemicals, particularly barium.
There are even web sites, where people who are convinced this theory is correct are taking the highly laudable step of attempting to verify their hypotheses with scientific experiments.
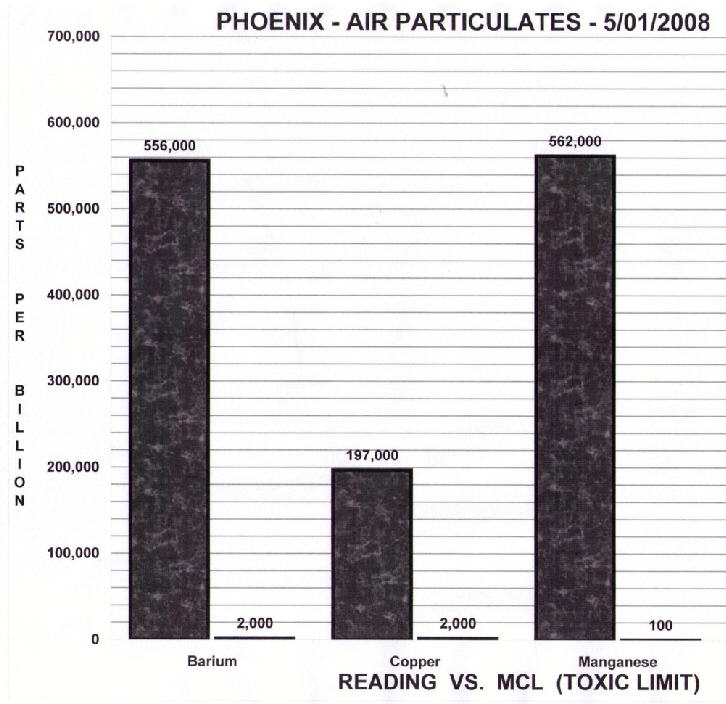
Unfortunately, they get the science terribly and inexcusably wrong. Take, for example, this graph found on the Arizona Skywatch site:
In particular, look at the first column. It shows a concentration of barium in the air of 556,000 ppb (parts per billion), vs. a “Toxic Limit” of 2,000 ppb. Clearly something is terribly wrong, we’ve got nearly 600 times the toxic limit of Barium in the air. Why are people not dropping dead all over Phoenix?
The first mistake is the value used for “Toxic Limit”. 2000 ppb is actually the EPS’s limit for barium in water, not in air. The limit for barium in air is 5 mg/m3 according to the OSHA. What’s that in ppb? Well, one m3 (cubic meter) of air weighs 1.2 kg, so that’s about 4000 ppb. Not significantly different from 2000 ppb, but as we’ll see. The fact that we are measuring the concentration in air is very important.
Still, 300 times over the toxic limit? And these limits were actually set for people who worked in barium plants. How much barium would you expect to find in the air in a normal city? The CDC’s has done some research:
Background levels of barium in the environment are very low. The air that most people breathe contains about 0.0015 parts of barium per billion parts of air (ppb). The air around factories that release barium compounds into the air has about 0.33 ppb or less of barium. Most surface water and public water supplies contain on average 30 parts of barium per billion parts of water (ppb) or less, but can average as high as 300 ppb in some regions of the United States. In some areas that have underground water wells, drinking water may contain more barium than the 2000 ppb limit set by EPA. The highest amount measured from these water wells has been 10,000 ppb. The amount of barium found in soil ranges from about 15,000 to 3,500,000 ppb.
The above paragraph has been modified to convert all the units to ppb. The original mixed ppm and ppb, which can be confusing.
But there’s a scary thing: the air that most people breath is 0.0015 ppb barium. That means the level found in Phoenix (556,000 ppb) is 370,000,000 times the normal amount!
So what’s going on here? What’s up with these crazy figures?
It turns out the answer is quite simple: they did not actually measure the concentration of barium in the air, they measured the concentration of barium in the airborne particulates.
That’s right, they measured how much barium there is in outdoor dust and smog. In the countryside the airborne dust has a lot of soil in it (especially with the noted “strong Arizona winds and dust storms), and in the city it also has industrial emissions. The amount of barium found is right in the normal range for soil (15,000 – 3,500,000 ppb), and being from a city, you’d also expect some barium emissions in there to bump up the figure. In short, these results are well within the normal (and safe) ranges.
But how did this mistake come about?
To measure the concentration of a particulate substance in air, you can do just what SkyWatch did – you run a vacuum cleaner for a while with a clean air filter, and then you collect the particulate matter from the filter and analyse it’s composition. You then do the one thing missing here: you multiply the result by the weight of the particulate sample divided by the weight of the air the sample was extracted from.
Air weighs 1.2Kg per cubic meter a cubic meter is 1000 liters. A vacuum cleaner can pull through
80 liters per minute, meaning we would get 1kg or air in about ten minutes, or 6 kg in an hour. But how exactly was the sample collected? See here:
http://d1027732.mydomainwebhost.com/charts/our%20charts/index.html
We also took air samples in Phoenix, on non-windy days, using a HEPA filter, then vacuuming the particulates from that filter into another pristine HEPA filter, which we then emptied into a sterile container.
Unfortunately they neglect to say how long they vacuumed for, but it must have been long enough to get some measurable amount of particulates on the filter. So let’s say an hour, and they got one gram (a pinch of salt worth).
So that would be 6Kg of air, meaning, with a 1g sample, we need to adjust our figures by a factor or 1/6000, which bring the claimed 556,000 ppb to 92 ppb, much closer to our normal range, and much lower than the OSHA limit of 4000 ppb.
So, bravo to Arizona Skywatch for trying to put some science behind their claims. But unfortunately they were not measuring what they though they were, and unless they can better describe their collection method, including how long they ran the vacuum for, and how much the submitted sample weighed, then their results are essentially meaningless, and show nothing more than normal air particulates.
So, how long will it take them to correct their site?
[UPDATE]
From here:
http://www.davidicke.com/forum/showpost.php?p=380613&postcount=55
This is the final Lab Report RE: air material analysis. This was a 28 day collection via HEPA filter, 8 hours per day collection. Please note the extremely high Aluminum reading 12,800,000 ppb.
The actual lab report is below.
Please let us know if you have research material surrounding this ‘report’. All ideas are encouraged. Previous analysis records are rain, surface water and soil.
Bridget Conroy
ArizonaSkyWatch.com
6-8-8
So that’s 28x8x6= 1,344 Kg of air (for an average vacuum cleaner). Now the only missing information was the weight of the particulates were in the hepa filter.
[UPDATE Jan 2010] When I wrote the above in 2008 I thought that Arizona Skywatch would at least remove the results from their page. But instead they left them there for a year, and then, unbelievably, they repeated the tests using the exact same methodology with the same results.:
http://d1027732.mydomainwebhost.com/charts/our%20charts/phx_particulates_2009.htm
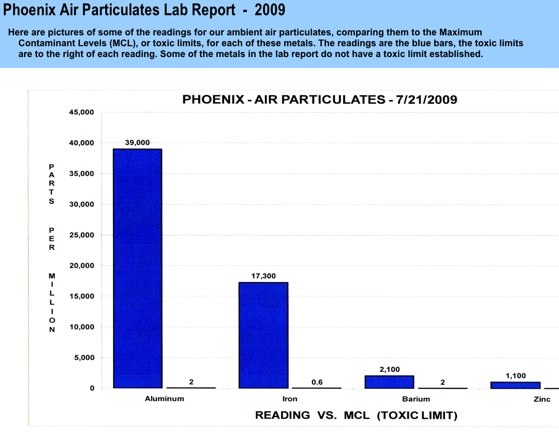
Incredibly bad science.

I reckon its cheaper to add stuff into your water. Why spray expensive chemicals in the air, when you can dose em on the ground…. Comtrails contrails conspiracy makes very little sense….
I just want to know why my trees are dead and dying.We are talking lots of trees and not only my trees they are like this everywhere in middle TN and the explaination in “Why in the World are They Spraying “makes sense.
no it doesn’t – if “chemtrails” were to blame then all trees EVERYWHERE would be dying – because whatever the poison is would be EVERYWHERE. Not just TN – but over the whole world!
I bet there are some trees within sight of yours that are not dying.
If you want to know why your trees are dying the first palce to ask would be an arborist – a tree specialist.
Unfortunately mass tree die-offs are not unheard of – for example in Europe they suffer “Ash dieback” – where 90% of Ash trrees can be killed by a known disease – see http://www.bbc.co.uk/news/world-europe-jersey-20501970 and http://en.wikipedia.org/wiki/Ash_dieback
windy and Mike,
This part of the country had a very hard year. I’ve been doing fall cleanup and the brush isn’t half of what it usually is. Drought hit hard this year. It’s not a big secret or mystery. As you said, all the arborists know.
Here is the report from July:
http://www.timesnews.net/article/9049134/drought-stress-on-trees-in-tennessee-deeper-than-is-evident
windy,
why would the ‘explanation’ in WhyITWATS make more sense than the prediction of climate change (likely caused by burning of fossile fuels)?
What we are seeing matches the projections of climate scientists. No conspiracy required.